by Boehringer Ingelheim /August 31, 2021
ENTERISOL SALMONELLA T/C® Vaccine Confers Protection Against Most Common Salmonella in Swine Clinical Cases

The monophasic Salmonella I 4,[5],12:i:- has emerged as the predominant Salmonella serovar isolated from clinical cases in the U.S. swine population.1,2 Infection can cause clinical disease in pigs and pose a public health threat.1,2 ENTERISOL SALMONELLA T/C® is the first and only U.S. commercial vaccine to have demonstrated protection against the new monophasic Salmonella serotype.2,3
STUDY KEY FINDINGS
- Pigs vaccinated with ENTERISOL SALMONELLA T/C® and challenged with monophasic Salmonella showed a significant (p < 0.05) improvement in average daily gain (ADG).3
- Vaccination led to a significant (p < 0.05) reduction in the occurrence of diarrhea.3
- Vaccination led to a significant (p < 0.05) reduction of intestinal lesions.3
ENTERISOL SALMONELLA T/C® is the first and only commercially available vaccine in the United States to have demonstrated efficacy in protecting against the most prevalent Salmonella serovar in the U.S. swine population.1,2,3
STUDY DESIGN
Overview
The study was conducted to determine if ENTERISOL SALMONELLA T/C® (the only commercially available vaccine in the U.S. that contains both a live Typhimurium serovar vaccine strain and a Choleraesuis serovar vaccine strain)4 confers protection against the predominant Salmonella serovar when isolated from clinical cases in the U.S. swine population. Vaccine efficacy was investigated by measuring clinical signs, intestinal lesions and ADG.3
Study Design
A randomized, controlled, blinded study evaluated pigs in three different treatment groups, each of them consisting of 20 pigs, blocked for the effects of litter, sex and weight: 1) non-vaccinated challenged (NVC); 2) non-vaccinated non-challenged (NVNC); and 3) vaccinated with ENTERISOL SALMONELLA T/C® and challenged (EVC). Animals were challenged four weeks post vaccination at 7 weeks of age with a dose of 2x109 S. I 4,[5],12:i:-. Pigs were monitored for clinical signs and enteric lesions and evaluated for weight gain for 14 days following the challenge.3
STUDY RESULTS
Results Summary
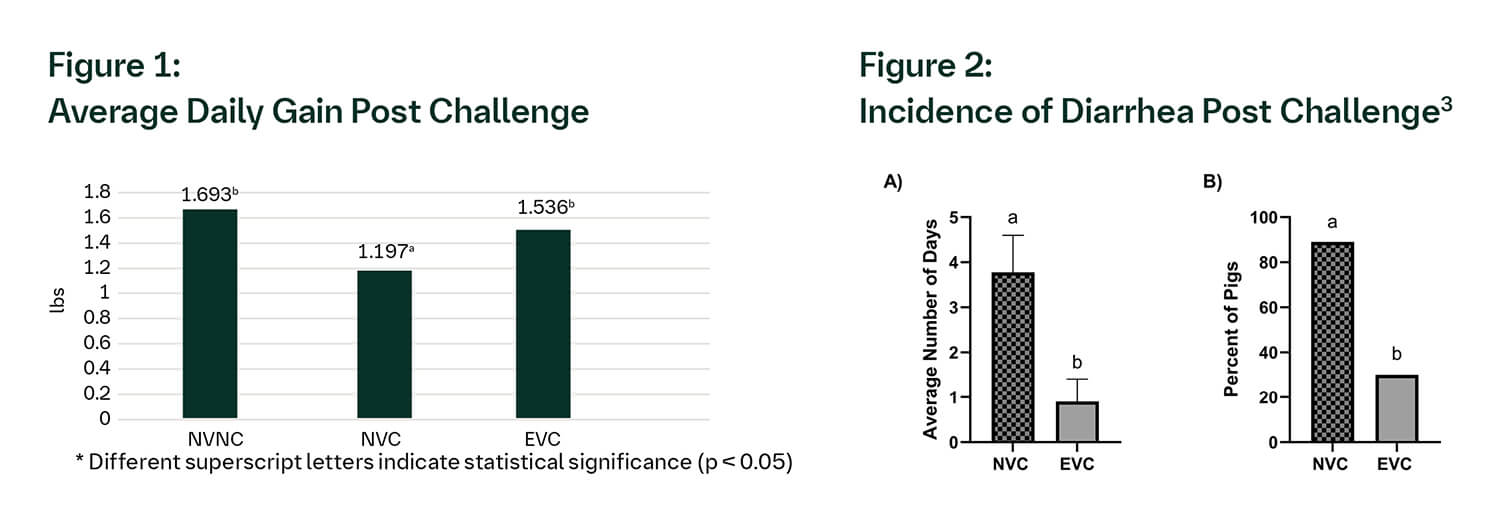
Animals in the EVC and NVNC groups had significantly (p < 0.05) greater ADG when compared with the NVC pigs (Figure 1). The EVC group had a significant (p < 0.05) decrease in both the duration of diarrhea as well as the percentage of pigs with diarrhea (Fig. 2A, Fig. 2B). One pig in the NVC group died due to salmonellosis; no mortality was found in the NVNC and EVC groups. Finally, four pigs in the NVC group had intestinal lesions, while no pigs had lesions in the NVNC and EVC groups.3
Conclusion
The results of this study show that the ENTERISOL SALMONELLA T/C® vaccine conferred protection against monophasic Salmonella as measured by clinical signs, intestinal lesions and improved average daily weight gain.3
ENTERSIOL SALMONELLA T/C® is the first and only commercially available vaccine in the United States to have demonstrated efficacy in protecting against the monophasic Salmonella serovar.2,3
References
1 Naberhaus SA, Krull AC, Bradner LK, et al. Emergence of Salmonella enterica serovar 4,[5],12:i:- as the primary serovar identified from swine clinical samples and development of a multiplex real-time PCR for improved Salmonella serovar-level identification. J Vet Diagn Invest 2019;31(6):818–827.
2 Salmonella enterica subsp. enterica I 4,[5],12:i:- Swine Health Information Center. 2021.
3 Leite F, Arruda P, et al. Oral live bivalent Salmonella vaccine reduces clinical disease, colonization and fecal shedding of multidrug resistant Salmonella enterica serovar I 4,[5],12:i:-. Vaccine, 2025.
ENTERISOL® and ENTERISOL SALMONELLA T/C® are registered trademarks of Boehringer Ingelheim Vetmedica GmbH, used under license. ©2025 Boehringer Ingelheim Animal Health USA Inc., Duluth, GA. All rights reserved. US-SWN-0028-2025