With an easy-to-use applicator, NexGard COMBO is made to ensure all product is administered and designed for fast application.
NexGard® COMBO topical solution offers one broad-spectrum solution that protects against fleas and ticks, prevents heartworm disease, and treats & controls roundworms, hookworms, and tapeworms.
With an easy-to-use plunger applicator, NexGard® COMBO topical solution is made to ensure all product is administered, designed for fast application and added confidence. Dosing is easy as 1,2,3!
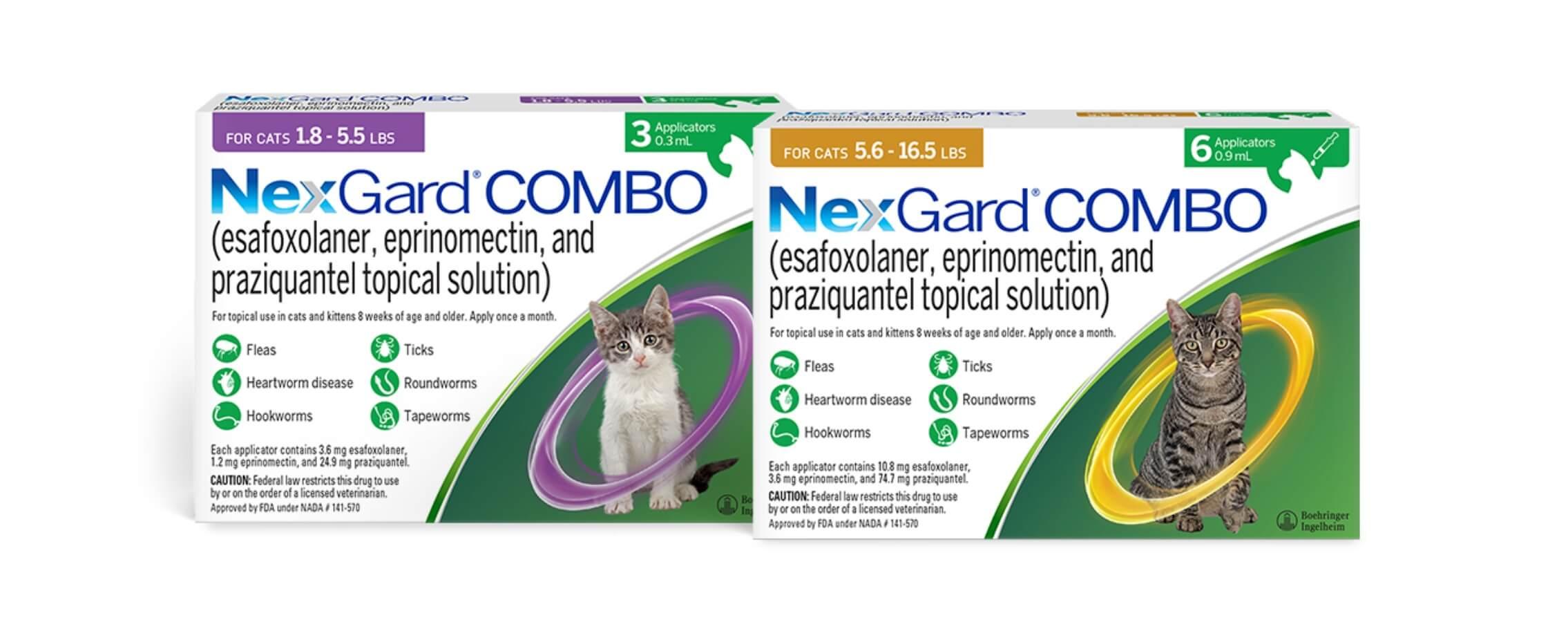
NexGard® COMBO topical solution combines the power of 3 active ingredients: esafoxolaner (designed specifically for felines), eprinomectin, and praziquantel. NexGard® COMBO topical solution is the first-ever broad-spectrum, isoxazoline-based parasiticide for cats that is indicated to treat tapeworms.
NexGard® COMBO (esafoxolaner, eprinomectin, and praziquantel topical solution) provides powerful protection for your feline patients. It is the first and only topical, broad-spectrum parasite treatment that protects against fleas and ticks, prevents heartworm disease, and treats and controls roundworms, hookworms and tapeworms, in a single monthly topical application.
NexGard® COMBO topical solution is safe for felines 8 weeks of age and older, weighing 1.8 lbs or more.
NexGard® COMBO topical solution is indicated for the prevention of heartworm disease caused by Dirofilaria immitis. NexGard® COMBO kills adult Ctenocephalides felis fleas and is indicated for the treatment and prevention of flea infestations, the treatment and control of black-legged tick (Ixodes scapularis) and lone star tick (Amblyomma americanum) infestations, and the treatment and control of roundworms (fourth-stage larval and adult Toxocara cati), hookworms (fourth-stage larval and adult Ancylostoma tubaeforme; adult Ancylostoma braziliense), and tapeworms (Dipylidium caninum).
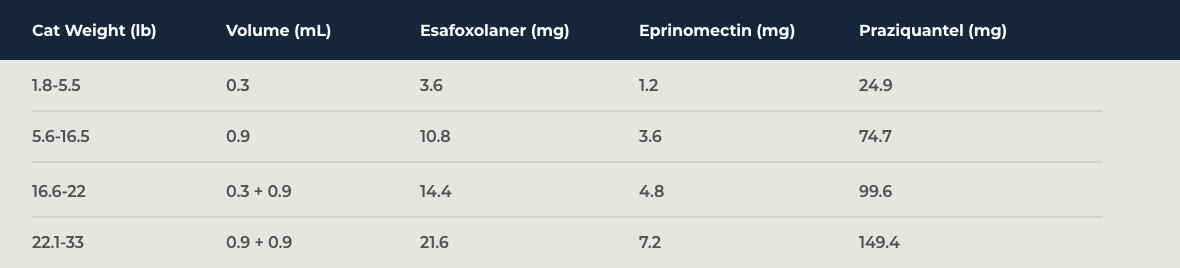
NexGard® COMBO topical solution is dosed at a minimum of 0.055 mL/lb (0.12 mL/kg), which delivers a minimum dose of 0.65 mg/lb (1.44 mg/kg) esafoxolaner, 0.22 mg/lb (0.48 mg/kg) eprinomectin, and 4.53 mg/lb (9.98 mg/kg) praziquantel.
For heartworm disease prevention, apply once monthly for at least 3 months after last exposure to mosquitoes. Administer the entire contents of a
NexGard® COMBO unit applicator topically once a month as specified in the following table:
Dosing Schedule
To apply NexGard® COMBO topical solution, use scissors to cut the blister along the dotted line, then pull the lid away. Remove the applicator from the package and hold it upright. Pull back the plunger slightly, then twist and pull off the cap. Part the hair on the midline of the neck, between the base of the skull and the shoulder blades until the skin is visible. Place the tip of the applicator on the skin and apply the entire contents directly onto the skin in one spot. The product should be applied to dry skin on an area where the cat cannot lick it off. If the weight of the cat requires a second application, apply the contents in the same manner as described above in the same location. Wash hands after use with soap and water.
Do not administer orally. Cats may salivate excessively if NexGard® COMBO topical solution is accidentally administered orally or is ingested through licking/grooming the application site (see Target Animal Safety). Esafoxolaner, one of the ingredients in NexGard® COMBO, is a member of the isoxazoline class. This class has been associated with neurologic adverse reactions including tremors, ataxia, and seizures. Seizures have been reported in cats receiving isoxazoline class drugs, even in cats without a history of seizures. Use with caution in cats with a history of seizures or neurologic disorders. The safety of NexGard® COMBO has not been fully evaluated in breeding, pregnant, or lactating cats. The safety of NexGard® COMBO has not been tested in kittens less than 8 weeks of age or weighing less than 1.8 lbs. The most frequently reported adverse reactions include vomiting, application site reactions, and anorexia.
NexGard® is a registered trademark and NexGard® COMBO™ is a trademark of Boehringer Ingelheim Animal Health France, used under license. ©2026 Boehringer Ingelheim Animal Health USA Inc., Duluth, GA. All rights reserved.